Disease-Modifying MS Therapies Market Forecast 2026–2036: Market to Reach USD 63,372.5 Million by 2036 at 7.1% CAGR
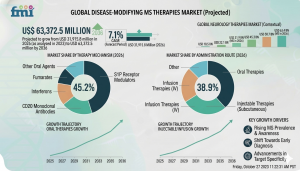
DE, UNITED STATES, March 17, 2026 /EINPresswire.com/ -- The disease-modifying MS therapies market is projected to be valued at USD 31,915.8 million in 2026 and expand to USD 63,372.5 million by 2036, reflecting a steady CAGR of 7.1% over the forecast period. This sustained value growth stems from the structural emphasis on long-term immune modulation in multiple sclerosis (MS) management, with therapies prescribed continuously over decades rather than episodically. Earlier treatment initiation, improved persistence, and prolonged exposure per patient are concentrating spending in chronic care settings, particularly in hospital and specialty neurology environments that support initiation, escalation, and ongoing safety oversight.
Market snapshot: global market 2026 - 2036
Market size 2026? USD 31,915.8 million
Market size 2036? USD 63,372.5 million
CAGR? 7.1%
Leading product category and share? Oral therapies (largest share among product categories)
Leading application and share? Relapsing-remitting MS (66.0%)
Leading end user and share? Hospital pharmacies (46.0%)
Key growth regions? Fastest-growing countries include India, China, Brazil, USA, and UK
Top companies? Biogen, Roche, Novartis, Sanofi, Merck KGaA
Market Momentum (YoY Path)
The disease-modifying MS therapies market follows a consistent upward trajectory driven by patient identification, early intervention, and therapy continuity. Starting from USD 31,915.8 million in 2026, the market builds steadily through improved diagnostic reach and sustained treatment adherence. By 2028 and 2030, cumulative exposure and broader neurology access contribute to incremental gains. Momentum continues into 2031 and 2033 as real-world evidence reinforces formulary positioning and long-term outcomes. The path culminates at USD 63,372.5 million by 2035–2036, supported by prolonged per-patient therapy duration and structural shifts toward sustained relapse reduction and disability delay.
Why the Market is Growing
Demand for disease-modifying MS therapies is rising as neurologists prioritize early intervention to slow progression, reduce relapse frequency, and preserve neurological function soon after diagnosis. Advances in MS pathophysiology understanding have expanded mechanism-based options, enabling more tailored selection based on disease subtype, activity, and patient tolerance. Growing MS prevalence—fueled by better diagnostics and broader screening—reinforces uptake of therapies aligned with guidelines focused on sustained relapse reduction and brain lesion suppression. Payers increasingly weigh real-world evidence and long-term disability outcomes in reimbursement, supporting access to agents showing durable benefits across diverse populations.
Drivers, Opportunities, Trends, Challenges
Drivers
Key drivers include early intervention strategies that target immune pathways to modify disease course, reduce relapses, and delay disability. Improved diagnostic accuracy, broader neurologist access, and guideline emphasis on sustained treatment expand the treated population and reinforce therapy persistence across decades.
Opportunities
Opportunities arise from expanding access in emerging systems through insurance coverage, domestic supply improvements, and neurology infrastructure growth. Oral formulations and reimbursement inclusion enhance adherence and utilization in high-potential markets, while real-world evidence strengthens positioning for long-term outcomes.
Trends
Trends reflect a shift toward high-efficacy agents like monoclonal antibodies for aggressive disease, alongside preference for oral therapies that ease treatment burden. Treatment sequencing balances efficacy, safety, and convenience over multi-year horizons, with monitoring and imaging guiding individualized plans.
Challenges
Challenges center on safety monitoring intensity (infection, malignancy risks), reimbursement thresholds, and biologic manufacturing complexity, which constrain rapid uptake of high-efficacy options. Access varies by healthcare maturity, while high costs, adverse event discontinuations, and specialist follow-up requirements limit scalability in some regions.
Competitive Landscape
The disease-modifying MS therapies market features established leaders evaluated on relapse reduction efficacy, progression delay evidence, safety burden, administration route, and long-term data. Biogen maintains a strong position with a broad portfolio spanning injectables, oral therapies, and infusions. Roche drives adoption through high-efficacy monoclonal antibodies in relapsing and progressive segments. Novartis emphasizes convenience and sustained outcomes via oral and infusion options. Sanofi focuses on immunomodulatory treatments for specific subgroups, while Merck KGaA supports demand with oral agents backed by established clinical use and global access. Differentiation hinges on durable efficacy, safety management, reimbursement negotiations, and alignment with evolving treatment algorithms.
Subscribe for Year-Round Insights → Stay ahead with quarterly and annual data updates: https://www.futuremarketinsights.com/reports/sample/rep-gb-31199
FAQ
What is the projected size of the disease-modifying MS therapies market in 2026 and 2036? The market is valued at USD 31,915.8 million in 2026 and forecast to reach USD 63,372.5 million by 2036.
What is the forecast CAGR for the disease-modifying MS therapies market from 2026 to 2036? The market is expected to grow at a CAGR of 7.1% during the period.
Which product category holds the largest share? Oral therapies represent the leading share due to convenient administration that supports long-term adherence and outpatient suitability.
Which application segment dominates demand? Relapsing-remitting MS accounts for 66.0% share, driven by high diagnosis rates and guideline-supported early treatment to reduce relapses and lesions.
Which end user setting leads in share? Hospital pharmacies hold 46.0% share, managing therapy initiation, infusions, and intensive monitoring for advanced protocols.
Which countries are experiencing the fastest growth? India (8.8%), China (8.6%), Brazil (8.3%), USA (7.7%), and UK (6.6%) show the strongest expansion, fueled by improved access, diagnosis, and care continuity.
Who are the top players in the disease-modifying MS therapies market? Leading companies include Biogen, Roche, Novartis, Sanofi, and Merck KGaA.
Explore More Related Studies Published by FMI Research:
Disposable Laparoscopic Instruments Market: https://www.futuremarketinsights.com/reports/disposable-laparoscopic-instruments-market
Corneal Ulcer Treatment Market: https://www.futuremarketinsights.com/reports/corneal-ulcer-treatment-market
IV Pole Market: https://www.futuremarketinsights.com/reports/iv-poles-market
Radiopharmaceutical Market: https://www.futuremarketinsights.com/reports/radiopharmaceuticals-market
Intraosseous Infusion Devices Market: https://www.futuremarketinsights.com/reports/intraosseous-infusion-devices-market
About Future Market Insights (FMI)
Future Market Insights, Inc. (FMI) is an ESOMAR-certified, ISO 9001:2015 market research and consulting organization, trusted by Fortune 500 clients and global enterprises. With operations in the U.S., UK, India, and Dubai, FMI provides data-backed insights and strategic intelligence across 30+ industries and 1200 markets worldwide.
Why FMI: Decisions that Change Outcomes- https://www.futuremarketinsights.com/why-fmi
Contact Us:
Future Market Insights Inc.
Christiana Corporate, 200 Continental Drive,
Suite 401, Newark, Delaware – 19713, USA
T: +1-347-918-3531
Website: https://www.futuremarketinsights.com
LinkedIn| Twitter| Blogs | YouTube
Have a specific Requirements and Need Assistant on Report Pricing or Limited Budget please contact us - sales@futuremarketinsights.com
Sudip Saha
Future Market Insights Inc.
+1 347-918-3531
email us here
Legal Disclaimer:
EIN Presswire provides this news content "as is" without warranty of any kind. We do not accept any responsibility or liability for the accuracy, content, images, videos, licenses, completeness, legality, or reliability of the information contained in this article. If you have any complaints or copyright issues related to this article, kindly contact the author above.